一、 巴西ANVISA监管信息简介

1. 监管局及简介
巴西卫生监管局(ANVISA, Agência Nacional de Vigilância Sanitária )是一个与卫生部有关的自治机构,是巴西国家卫生系统(SUS)的一部分,是巴西卫生监管系统(SNVS)的协调者,在全国范围内存在。ANVISA的作用是通过对受卫生监管的产品和服务的生产、销售和使用进行卫生控制,包括相关的环境、流程、成分和技术,以及港口、机场和边境的控制,促进对人口健康的保护。
2. 监管法规
• Medical Equipments: Resolution RDC 185/2001 and RDC 40/2015
• Materials for health use: Resolution RDC 185/2001 and RDC 40/2015
• Orthopedic Implants: Resolution RDC 185/2001
• In vitro diagnostics: Resolution RDC 36/2015
• Clinical Trials 临床试验:The regulation that normalizes the submission of clinical trials with Medical Devices to ANVISA’s approval is the Resolution RDC 10/2015
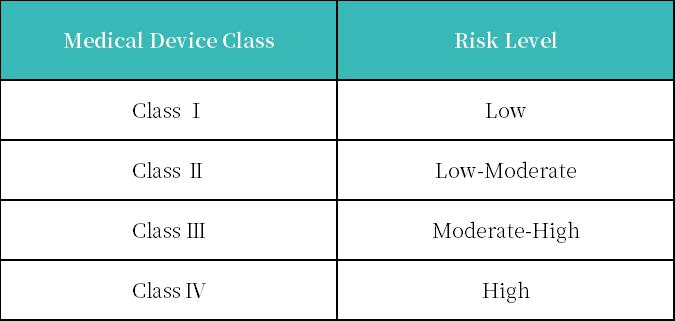
3. 医疗器械和IVD产品风险等级分类
Anvisa 将医疗设备分为四类:医疗设备、卫生材料、整形外科植入物和体外诊断。
巴西的医疗器械根据其对人体构成的风险分为四类。
巴西最新法规是RDC 751/2022,已于2023年3月1日生效,新法规更贴近欧盟,包括医疗器械定义,分类规则。医疗器械也遵循22个分类规则,这些规则在很大程度上类似于欧洲医疗器械指令(MDD)93/42/EEC中概述的22个规则。
4. 准入必要条件
在巴西没有实际地点的外国医疗器械制造商必须指定一个巴西注册持有人 (BRH)。BRH作为制造商公司和巴西国家卫生监督局(ANVISA) 之间的联络人,负责制造商在巴西的医疗器械注册,并在设备注册上注明。
制造商只要在巴西销售医疗器械,就必须保持BRH。
巴西注册持有人将负责在其有效期内维护注册,制造商及其指定经销商将独家使用此记录和服务。
5. 注册语言
英语+葡萄牙语
二、 注册流程,周期&官费
1. 注册流程图

2. 注册路径
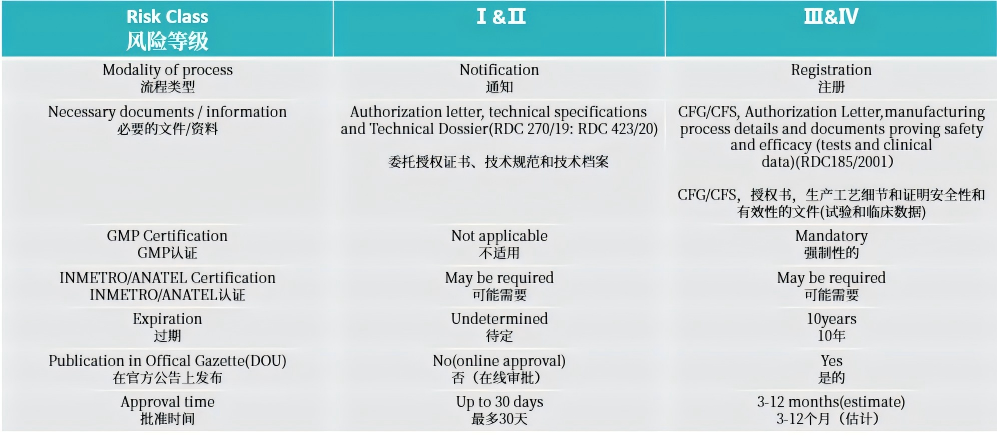

3. 注册周期及官费

三、 FAQ
Q:提交注册资料后有几次发补的机会?
A:医疗器械注册过程受程序评估、重新评估、审计、市场监测和检查。若发现不一致或需补充信息,持有人需在 30 天内调整;对先前提交信息的更正需通过特定申请处理;若 30 天内未对调整通知做出回应,可能导致注册被取消。
Q:对于BRH要求是什么?
A:所有在巴西没有实体办公场所的制造商都必须任命一名巴代作为BRH,因为只有巴西本地的企业才可以申请产品注册,并且证书是由BRH持有。BRH可以是企业在巴西的商业合作伙伴、企业在巴西的分支机构、在巴西有分支机构的咨询机构。BRH必须持有营业许可AFE(Autorização de Funcionamento)的资质。
Q:在ANVISA注册医疗器械强制原产国注册吗?
A:不需要原产国注册。
Q:如果有GHTF注册记录是否可以加快?
A:如果有欧盟、美国、日本、澳大利亚和加拿大的批准,可以影响巴西当局的审查程序。
Q:制造商有MDSAP体系证书了,是不是就不需要申请BGMP?
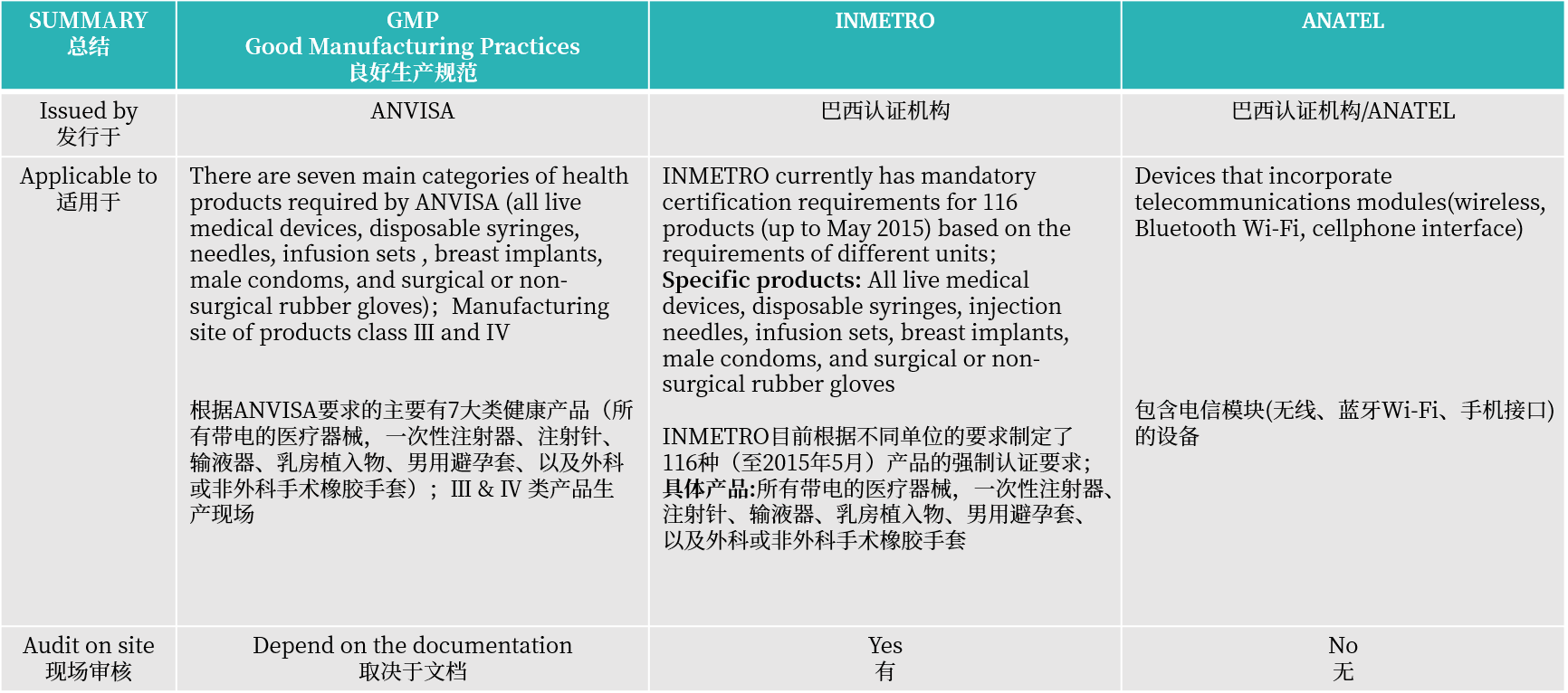
A:制造商必须在III类和IV类医疗器械的产品注册之前获得巴西良好生产规范(BGMP认证)。MDSAP证书只是加速GMP认证批准的一个很好的参考,但并不能取代它。如果有MDSAP证书可以只进行文件审核,极大可能免去BGMP现场审核。
Q:什么是INMETRO认证?
A:国家计量、标准化和工业质量研究所(INMETRO)对电子医疗设备的认证。证书有效期为无有效期,INMETRO认证由产品认证机构或OCP(如SGS、TÜV Rheinland、UL)进行。与医疗器械注册一样,INMETRO证书需要获得许可的BRH,如果没有巴西当地代表,外国制造商无法自行获得INMETRO认证。作为医疗器械注册的一部分,所有适用的产品必须在注册申请中提供其INMETRO证书的公证副本。
Q:注册证书可以转让吗?
A:根据RDC第102/2016号决议,所有权转让将被视为新的登记程序。
Q:巴西注册证书的有效期是多久?
A:ClassI&II永久有效;ClassIII&IV有效期10年。

